What is Stem Cell Therapy?
Conditions We Treat
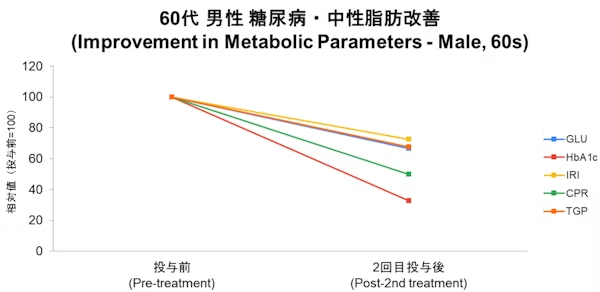
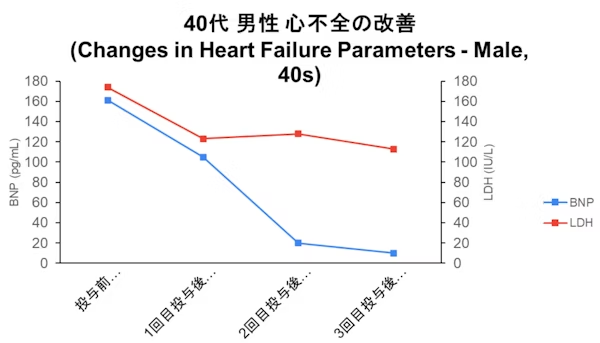
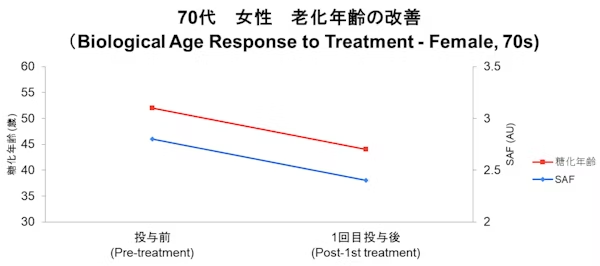
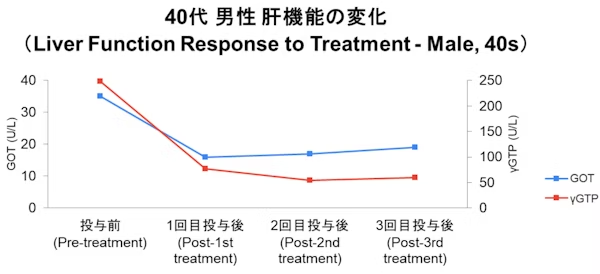
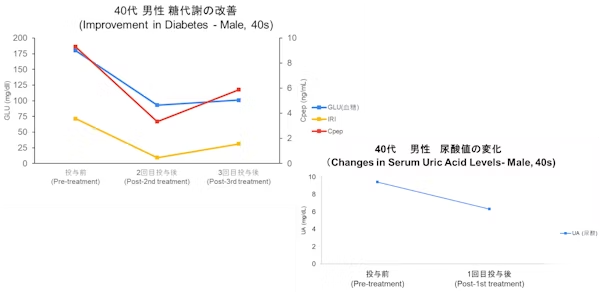
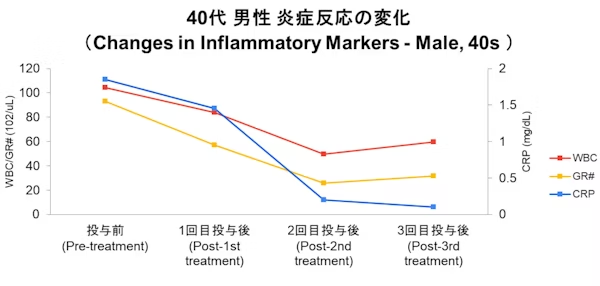
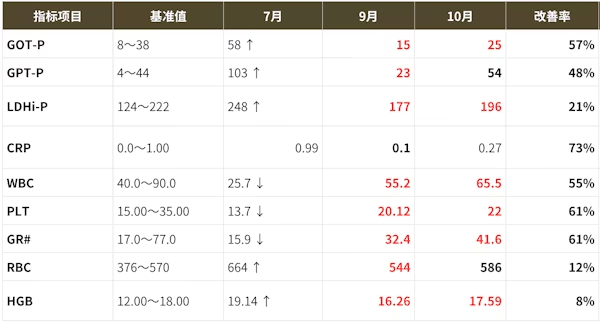
Case
Stem Cell Therapy Programs at Tokyo Relife Clinic
Why Choose Stem Cell Therapy?
Issues with Standard Stem Cell Therapy
Our Solution: How Tokyo Relife Clinic Addresses These Challenges
Typical Treatment Schedule for Stem Cell Therapy
Procedure Guide: Initial Fat Tissue Collection
Culture Period
What to Expect: Day of Stem Cell Infusion
Who May Not Be Suitable for Stem Cell Therapy
PRICE
Frequently Asked Questions (FAQ)
What is Stem Cell Therapy?
Stem cell therapy is a regenerative medicine approach that utilizes the tissue repair and immune modulation properties of stem cells to support the body's natural regenerative capabilities and anti-inflammatory responses. This treatment targets declining bodily functions caused by aging, chronic diseases, and lifestyle-related conditions (such as diabetes) at the cellular level to support fundamental repair and improvement.

In Japan, our treatment involves collecting stem cells from the patient's own adipose tissue, culturing them to reach the target quantity, and then administering them back into the body via infusion or local injection. Because we use only autologous cells (the patient's own cells), the risks of allergic reactions and immune rejection are relatively low.
Stem cells are known to continuously divide and differentiate while simultaneously secreting various biological factors※1. These factors play important roles in regulating inflammatory responses and supporting tissue repair※2. Through these mechanisms, various research studies suggest that stem cells may help maintain bodily functions or support improvement of chronic conditions. (Source:※1 Weissman IL, et al. Cell. 2000;100(1). ※2 Caplan AI, Dennis JE. J Cell Biochem. 2006;98(5).)
Conditions We Treat
Our stem cell therapy may be suitable for those with:
- Chronic pain
- Stroke (cerebrovascular disorders)
- Declining brain function
- Diabetes
- Menopausal / Andropausal symptoms
- Age-related physical decline (longevity)
- Liver disease
- Knee osteoarthritis (joint pain)
- Muscle, tendon, or ligament injuries
- Arteriosclerosis (lifestyle-related disease risks)
- Hair thinning or loss
- Erectile dysfunction (ED)
※Suitability determined by physician consultation.
Case
Case Study Disclaimer & Precautions
- Disclaimer:Case photos show specific patient outcomes at our clinic. Individual results vary and are not guaranteed. Outcomes depend on baseline condition, age, and lifestyle.
- Side Effects & Risks:Injection site pain/bruising, allergic reactions, infection risk, pulmonary embolism/phlebitis (extremely rare), unintended cell proliferation.
- Legal Notice:This treatment is provided under Japan's Act on the Safety of Regenerative Medicine and is not covered by insurance. Stem cells are processed by our certified partner facility.
Stem Cell Therapy Programs at Tokyo Relife Clinic
In Japan, the "Act on the Safety of Regenerative Medicine" was enacted in 2014 to ensure the safety of regenerative medicine treatments.
At Tokyo Relife Clinic, we provide stem cell therapy in full compliance with this Act. Our treatment protocols, cell handling procedures, and quality management systems have undergone rigorous regulatory review. We submit our Regenerative Medicine Treatment Plans to the appropriate authorities and have received official acceptance from the Ministry of Health, Labour and Welfare.
Currently, our clinic has obtained acceptance for nine (9) Type II Regenerative Medicine Treatment Plans.
Stem Cell Therapy: Regenerative Medicine Treatment Plan Acceptance Numbers
- PB3240265: Regenerative medicine using autologous adipose-derived stem cells for cerebrovascular disorders
- PB3240266: Regenerative medicine using autologous adipose-derived stem cells for glucose metabolism abnormalities
- PB3240267: Regenerative medicine using autologous adipose-derived stem cells for menopausal and andropause symptoms
- PB3240268: Regenerative medicine using autologous adipose-derived stem cells for liver dysfunction
- PB3240269: Regenerative medicine using autologous adipose-derived stem cells for frailty
- PB3240263: Regenerative medicine using autologous adipose-derived stem cells for chronic pain
- PB3240294: Regenerative medicine using autologous adipose-derived stem cells for hair tissue
- PB3250002: Regenerative medicine using autologous adipose-derived stem cells for erectile dysfunction
- PB3250003: Regenerative medicine using autologous adipose-derived stem cells for osteoarthritis









Why Choose Stem Cell Therapy?
Aging is understood to progress through multiple interconnected factors, including immune system aging, accumulation of senescent cells, chronic inflammation, DNA damage, and decline in stem cell function and quantity. These factors do not operate independently; rather, they interact with one another to contribute to age-related decline in bodily functions.
Under normal circumstances, stem cells play a vital role in maintaining and repairing tissues in response to daily cellular damage and aging. However, as we age, both the number and function of stem cells gradually decline. Research has shown that stem cell activity in middle-aged and older adults is significantly reduced compared to younger individuals (Source:López-Otín C, et al. Cell. 2013;153).

As a result, tissue repair takes longer, and various age-related changes become more apparent, including declining organ function, reduced skin elasticity, joint degeneration, and other manifestations of aging.(Source: Stolzing A, et al. Mech Ageing Dev. 2008;129(3).)
This diagram is a schematic representation showing the trend of declining stem cell quantity and function with aging.
Stem cell therapy is a regenerative medicine approach that addresses the age-related "decline in stem cell quantity and function" underlying the aging process. This treatment aims to support the body's internal environment regulation and enhance its natural recovery capabilities.
Issues with Standard Stem Cell Therapy
■Lack of Scientific Evidence
Some clinics perform treatments without scientific evidence due to a lack of specialized knowledge.
■Unclear Treatment Effects
The criteria for evaluation are subjective, making the effectiveness unclear.
■Unclear Quality and Quantity of Cells Administered
There are no objective standards for assessing the cells administered, making it difficult to maintain consistent quality.
Our Solution: How Tokyo Relife Clinic Addresses These Challenges
■Evidence-based Treatment
With the integration of medical facilities and research labs, we can conduct clinical research and quickly implement the results in the clinical setting, making evidence-based treatment possible.
■Proving Treatment Effects with Testing
We utilize epigenetic clock testing to scientifically measure your biological age at the DNA level. This assessment serves as a key objective indicator, allowing us to evaluate the efficacy of cellular treatments from multiple perspectives by comparing data before and after the procedure.
■Testing of Cells Before Administration
By having an in-house cell culture facility, cells can be cultured and administered with a single freeze, preventing degradation from freezing. Furthermore, the cultured cells are activated before administration and undergo various safety tests, ensuring both the quality and quantity of the cells.
Typical Treatment Schedule for Stem Cell Therapy
At Tokyo Relife Clinic, we recommend stem cell infusion intervals based on treatment objectives and individual patient conditions. Generally, during the initial treatment phase, we recommend three infusions administered every three months while monitoring changes in the body's internal environment and making necessary adjustments.
Following this initial phase, infusion intervals are adjusted in consultation with your physician based on disease progression and examination results.
Treatment Goal | Recommended Interval (After Initial 3 Sessions) |
|---|---|
Aesthetic & Anti-Aging Care(maintaining skin condition, etc.) | Every 3 to 6 months |
Joint-Related Conditions | Every 6 months to 1 year |
Chronic Conditions | Every 3 months to 1 year |
Neurological Conditions | Every 6 months to 1 year |
Erectile Dysfunction | Every 6 months to 1 year |
Scalp Environment & Hair Regeneration | Every 3 to 6 months |
These intervals are general guidelines only. Actual treatment schedules will be determined individually through physician consultation.
Procedure Guide: Initial Fat Tissue Collection

1. Medical Questionnaire & Consultation (Approximately 20 minutes)
First, you will complete a medical questionnaire to confirm your current health status, medical history, and any allergies. Our nurse will then discuss your specific needs and treatment goals in detail.

2. Physician Examination & Consent Form (Approximately 30 minutes)
The physician will conduct an examination and explain the scope of fat tissue collection procedures and the day's schedule. After reviewing the treatment plan and addressing any questions, you will be asked to sign the consent form.
3. Blood Collection & Laboratory Testing (Approximately 2 hours)
Blood tests will be performed to assess your overall health status and screen for infectious diseases to determine final suitability for the procedure. If you have submitted recent blood test results in advance, the procedure may proceed without waiting for new test results.
If there are discrepancies between submitted results and our clinic's test results, Tokyo Relife Clinic's results will take precedence.

4. Payment Processing (Approximately 10 minutes)
While waiting for blood test results, we will complete the payment procedures.

5. Fat Tissue Collection
After administering local anesthesia, a small amount of fat tissue will be collected primarily from the abdominal area. Based on the physician's assessment, collection may also occur from the thighs or buttocks.
You may choose between two collection methods: blocking method or suction method. Regardless of the method selected, the actual procedure time is approximately 15 to 20 minutes.
- Blocking Method: Approximately 1 cm incision to collect 0.1 to 5 g of fat tissue
- Suction Method: Approximately 0.5 cm incision to collect approximately 10 cc of fat tissue
Including pre-procedure preparation and post-procedure recovery time, the total time from entering to leaving the recovery room is approximately 40 to 60 minutes.
After the procedure, the incision will be sutured internally and protected externally with gauze. Suture removal is not required.
Culture Period
Under typical circumstances, it takes approximately 4 to 6 weeks to culture and expand the cells to the target quantity needed for treatment. The time required for cell culture is closely related to the activity status of the patient's own cells.
What to Expect: Day of Stem Cell Infusion

1. Medical Assessment & Physician Consultation
Upon arrival at the clinic, you will complete various medical assessment forms regarding your current health status, symptoms, and lifestyle. Following this, the physician will conduct a consultation and make the final determination regarding your suitability for stem cell infusion on that day.

2. Blood Collection for Future Culture & Infectious Disease Screening
For patients planning future stem cell infusions, blood will be collected for stem cell culture and infectious disease testing. Please note that depending on the infectious disease test results, stem cell culture may not be possible.
3. Stem Cell Infusion
Vital sign monitoring equipment will be attached to your body.
The infusion duration varies depending on the quantity of stem cells administered and individual patient conditions, typically ranging from approximately 90 to 180 minutes.
During the infusion, you will be connected to monitoring equipment such as electrocardiogram (ECG), and medical staff will continuously monitor your vital signs.

4. Post-Infusion Observation
After the infusion is completed, you will be observed for approximately 15 to 30 minutes. Once your condition is confirmed to be stable, you may return home.
Who May Not Be Suitable for Stem Cell Therapy
Stem cell therapy may not be appropriate for those who:
- Have experienced severe allergic reactions to anesthetic agents or specific medications in the past
- Test positive for pathogenic microorganism screening (HIV)
- Have a history of allergic reactions to amphotericin B, human albumin, streptomycin, gentamicin, or vancomycin
- Have concurrent malignant tumors
- Are pregnant, breastfeeding, or undergoing dialysis
- Are deemed unsuitable for this regenerative medicine treatment by the treating physician for any other medical reason
Additional precautions and considerations for stem cell therapy are detailed on our comprehensive guidelines page.
PRICE
IV DripStem Cell Therapy(Standard Ver.)
IV DripStem Cell Therapy (Premium Ver.)
IV DripRelife Stem Cell Therapy
Annual Storage Fee
Local InjectionED (Erectile Dysfunction) Treatment
Local InjectionOsteoarthritis Treatment(Knee)
Manual Injection・MesotherapyHair Follicle Tissue Treatment
References:
※1:Weissman IL, et al. Cell. 2000;100(1).
※2:Caplan AI, Dennis JE. J Cell Biochem. 2006;98(5).
※3:López-Otín C, et al. Cell. 2013;153
※4:Stolzing A, et al. Mech Ageing Dev. 2008;129(3).
※5:Pittenger MF, et al. Science. 1999;284(5411).
※6:Woodbury D, et al. J Neurosci Res. 2000;61(4).
※7:Wang J, et al. J Hepatol. 2006;45(4).
※8:Morigi M, et al. J Am Soc Nephrol. 2004;15(7).
※9:Tang DQ, et al. Diabetes. 2004;53(7).
※10:Wu Y, et al. Stem Cells. 2007;25(10).